References:
- Lau, J and Thompson, J (2023) Intraosseous Device Insertion for Adults and Pediatrics. Association for Vascular
Access. Available at: https://cdn.ymaws.com/www.avainfo.org/resource/resmgr/files/position_statements/ava_position_paper_-_intraos.pdf.
Accessed February 20, 2023.
- Davidoff J, Fowler R, Gordon D, et al. JEMS. 2005;30(10):s20-s23. Research sponsored by Teleflex Incorporated.
- Cooper BR, Mahoney PF, Hodgetts TJ, et al. J R Army Med Corps. 2007;153(4):314-316.
- Montez DF, et al. Ann Emerg Med. 2015;66(4s):S47. Research sponsored by Teleflex Incorporated.
- Puga T, Montez D, Philbeck T, et al. Crit Care Med, 2016; 44(12):143. Research sponsored by Teleflex Incorporated.
Based on a healthy volunteer study.
- Philbeck TE, et al. JEMS. 2010;35(9):58-69. Research sponsored by Teleflex Incorporated.
- Hoskins SL, Nascimento P Jr., Lima RM, Espana-Tenorio, JM, Kramer GC. Pharmacokinet`ics of
intraosseous and central venous drug delivery during cardiopulmonary resuscitation.
Resuscitation 2011; doi:10.1016/j.resuscitation.2011.07.041. Research sponsored by Teleflex
Incorporated. (preclinical study)
- Truemper EJ, et al. Ann Emerg Med. 2012;60(4):S90. Research sponsored by Teleflex Incorporated.
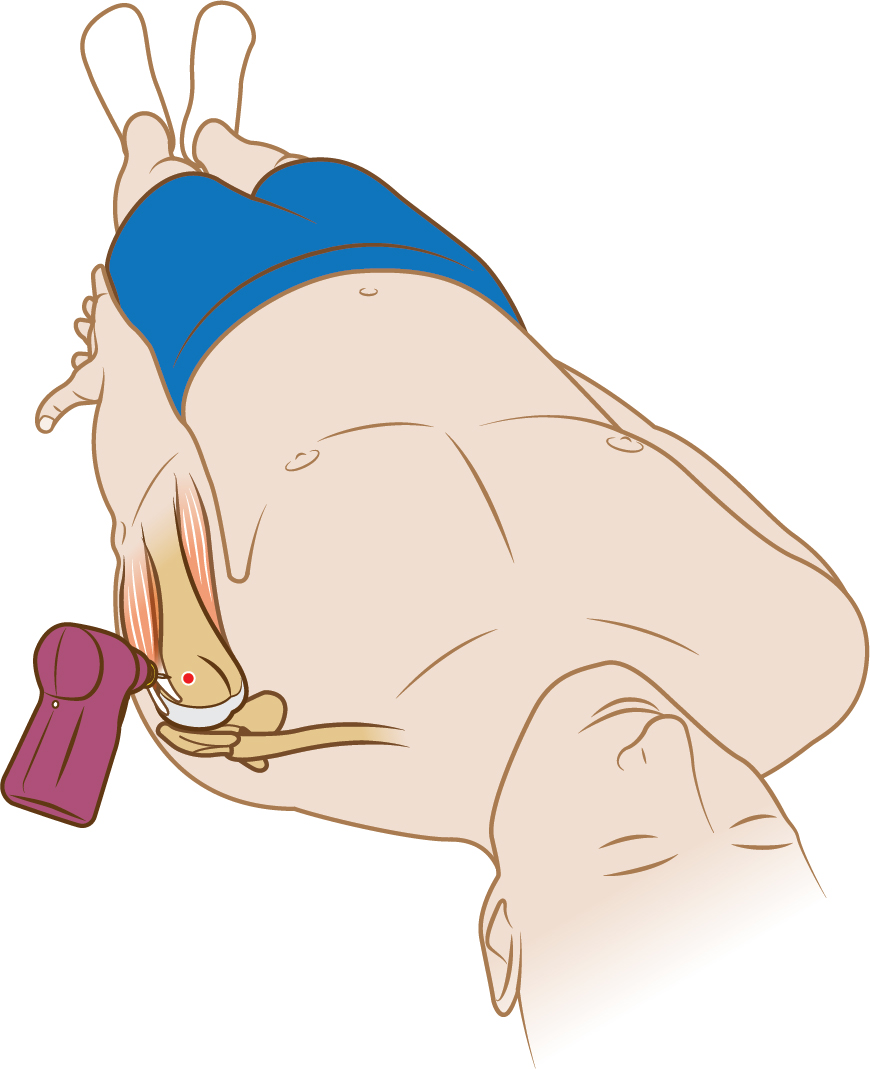
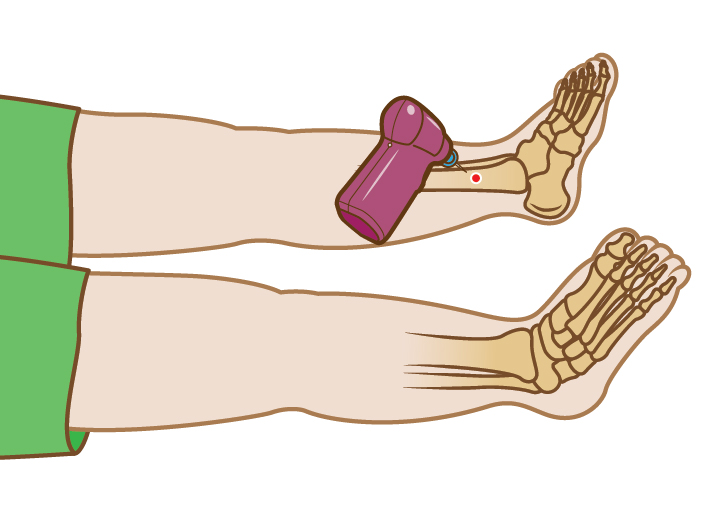
*The Arrow™ EZ-IO™ System is indicated for intraosseous access anytime in which
vascular access is difficult to obtain in emergent, urgent or medically
necessary cases for up to 24 hours. For patients ≥ 12 years old, the device may be extended for up to 48
hours when alternate intravenous access is not available or reliably established.
**This program has been approved by the American Association of Critical-Care Nurses (AACN), for 1.50 CERPs, Synergy
CERP Category A, File Number 23875. Approval refers to recognition of continuing education only and does not imply AACN
approval or endorsement of the content of this educational activity, or the products mentioned.
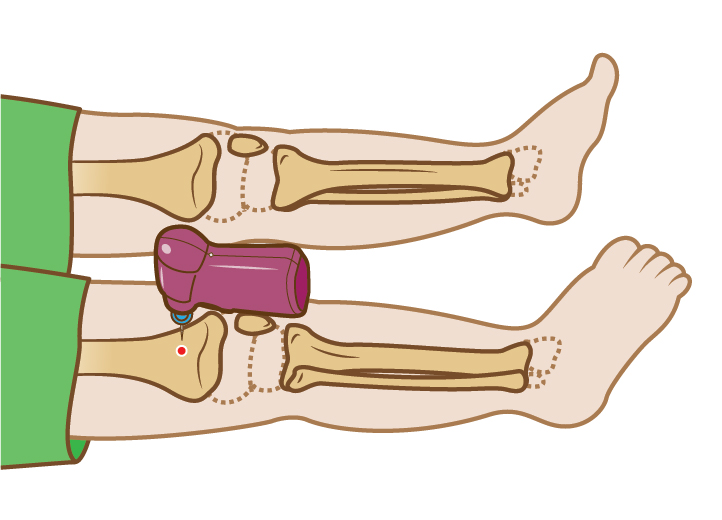
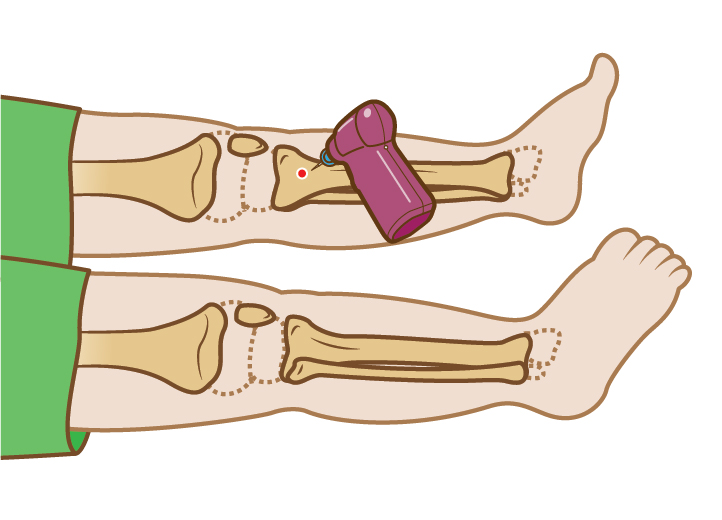
† Based on adult humerus data
§ Based on 300 mmHg of pressure
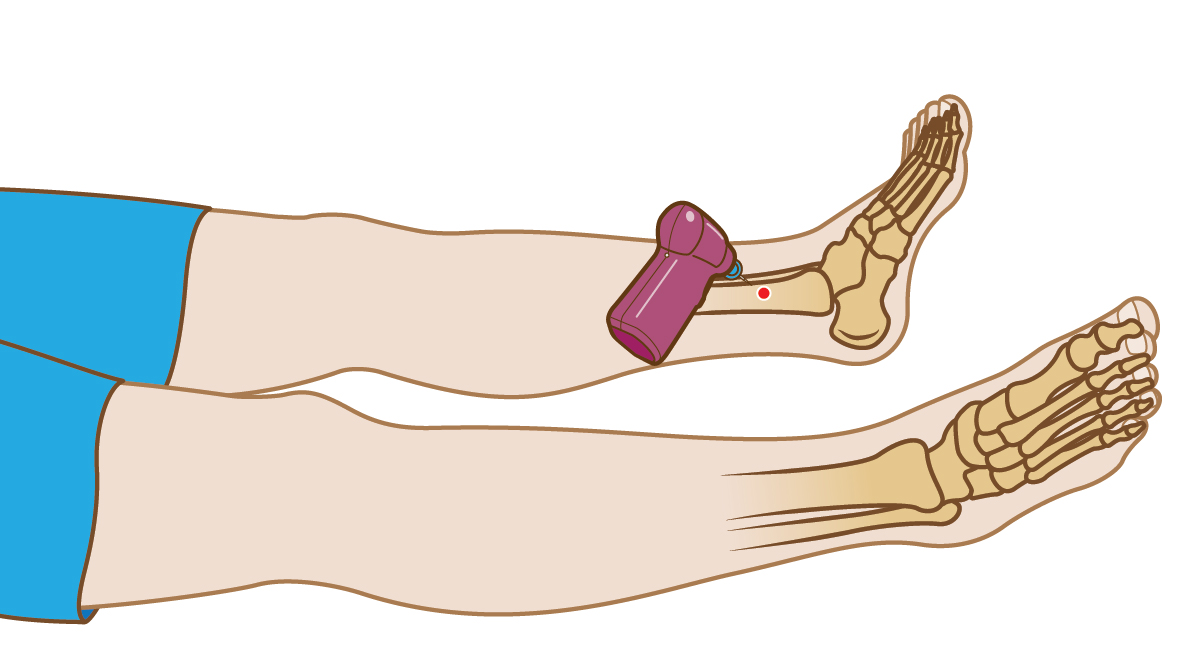
‡ Compared to EZ-IO™ System tibial insertions
|| Based on adult proximal tibial data and 300 mmHg of pressure
Rx Only.
CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician.
*The Arrow™ EZ-IO™ System is indicated for intraosseous access anytime in which
vascular access is difficult to obtain in emergent, urgent or medically necessary cases for up to 24 hours. For patients ≥
12 years old, the device may be extended for up to 48 hours when alternate intravenous access is not available or reliably
established. *Time to access is measured as insertion of the needle set through the bone cortex and into the intraosseous
space.
The Arrow™ EZ-IO™ Needle Set is Sterile, Single Use: Do not reuse, reprocess or
re-sterilize. Reuse of device creates a potential risk of serious injury and/or infection which may lead to death. Refer
to Instructions for Use for complete warnings, indications, contraindications, precautions, and potential complications.
Teleflex, the Teleflex logo, Arrow, EZ-IO, and The IO You Know are trademarks or registered trademarks of Teleflex
Incorporated or its affiliates, in the U.S. and/or other countries. All other trademarks or registered trademarks are the
property of their respective owners. MC-009068