Bioresorbable
Gel-Bead™ Embolization Spheres demonstrated degradation beginning at 4 weeks with some spheres still present at 12 weeks.*
4 Week Histology
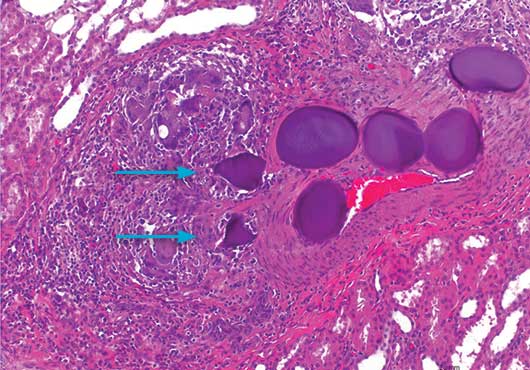 20x magnification, H&E stain
20x magnification, H&E stain
Gel-Bead™ 100-300μm Sphere implant site at 4 weeks. As shown above, Gel-Bead™ Spheres began to demonstrate significant degradation (arrows), mediated by the infiltration of inflammatory cells.
12 Week Histology
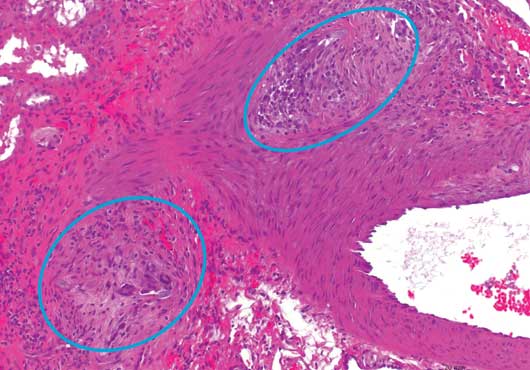 20x magnification, H&E stain
20x magnification, H&E stain
Gel-Bead™ 100-300μm Sphere implant site at 12 weeks. Tissue surrounding the presumed implant site consisted of fibrous connective tissue containing macrophages and multinucleated giant cells.
Bioresorbable
Spherical shape designed to provide consistent embolic delivery with predictable sphere size distribution.
Gelatin Spheres
Gelatin Slurry

For illustrative purposes only.
Multiple Size Ranges
Available in four size ranges for targeted embolization of hypervascular tumors.
